News
What Is an Optical Trocar? Function, Types & Comparison Guide
What Is an Optical Trocar? Function, Design, and Comparison with Classic and Bladeless Types
This article explains what an optical trocar is, how it enables real-time tissue visualization during laparoscopic access, how it compares to classic and bladeless trocars, and key procurement considerations for global medical distributors and OEM sourcing programs.
What Is an Optical Trocar?
An optical trocar is a specialized laparoscopic access device designed to allow visual confirmation of tissue layers during insertion. Unlike classic or bladeless trocars, an optical trocar incorporates a clear, transparent obturator that accommodates a laparoscopic scope — enabling the surgical team to observe tissue planes in real time as the trocar is advanced through the abdominal wall.
Optical trocars are used in minimally invasive surgery (MIS) and laparoscopic procedures where direct visualization during access placement is a procedural preference or clinical requirement.
As one of the three primary trocar types used in modern laparoscopic surgery — alongside classic and bladeless trocars — the optical trocar serves a distinct role within the laparoscopic access system.
How Does an Optical Trocar Work?
The defining feature of an optical trocar is its transparent or translucent obturator tip. This design allows a 0° laparoscope to be inserted through the trocar cannula during access, so the surgeon can directly visualize each tissue layer — skin, fascia, muscle, and peritoneum — as the device is advanced.
Step-by-Step Access Process
- A laparoscope is loaded into the transparent obturator of the optical trocar
- The trocar is positioned at the intended abdominal entry point
- As the device is advanced, the surgeon observes tissue layers through the scope in real time
- Once the peritoneal cavity is confirmed visually, the obturator is removed
- The cannula remains in place as the working access port
This visual feedback distinguishes optical trocars from other access methods, where tissue entry relies on tactile confirmation or pre-established anatomical landmarks alone.
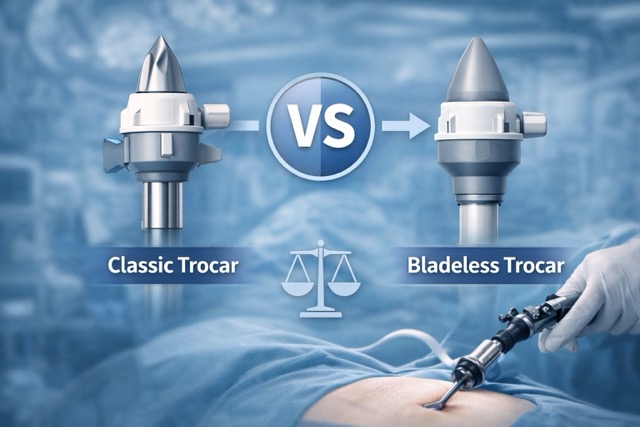
Optical Trocar vs Classic Trocar vs Bladeless Trocar
Understanding how optical trocars compare to the other two primary types helps procurement teams and clinical engineers select the right device for each workflow. For a detailed comparison of classic and bladeless designs, see: Bladeless Trocar vs Classic Trocar — Key Differences & Selection Guide .
| Feature | Classic Trocar | Bladeless Trocar | Optical Trocar |
|---|---|---|---|
| Obturator design | Traditional blade tip | Tissue-separating tip (no blade) | Transparent / clear tip |
| Visual confirmation | No | No | Yes — real-time tissue visualization |
| Scope compatibility | Not during insertion | Not during insertion | Yes — 0° laparoscope inserted during access |
| Tissue entry method | Puncture / cutting | Tissue separation (dilating) | Cutting or dilating with visual guidance |
| Typical use case | Routine standardized access | Tissue-sparing preference | Workflows requiring visual layer confirmation |
| Additional equipment needed | None beyond trocar | None beyond trocar | Laparoscope required during insertion |
When Are Optical Trocars Used in Laparoscopic Surgery?
Optical trocars are selected in laparoscopic workflows where visual feedback during abdominal access is a procedural preference. Common clinical contexts include:
Procedures with Anatomical Complexity
Where visual confirmation of tissue planes supports more controlled access placement.
Reoperative or Adhesion Cases
Where prior surgical history may have altered standard anatomical tissue relationships.
Institutional Protocol Requirements
Some surgical centers specify optical access as a standard for initial port placement.
Surgeon Workflow Preference
Surgeons trained with optical access techniques may specify this trocar type as a standard component of their laparoscopic setup.
Optical trocars are one component within the broader minimally invasive surgery (MIS) device ecosystem , working alongside suction irrigation systems, insufflation filters, specimen retrieval bags, and OR safety consumables.
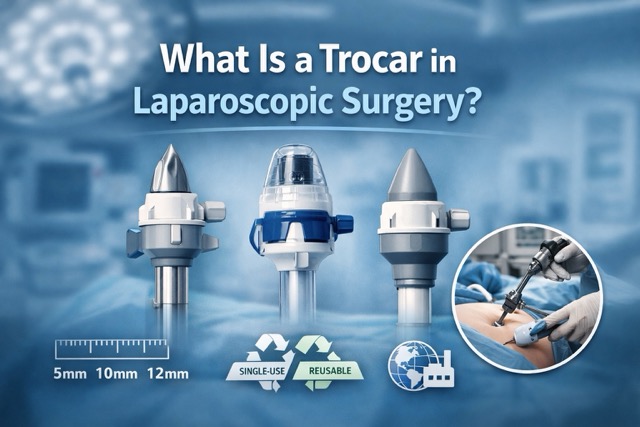
Optical Trocar Sizes and Configurations
Optical trocars are typically available in standardized diameters aligned with common laparoscopic instrument compatibility requirements:
- 5 mm — for smaller laparoscopic instruments requiring visual access confirmation
- 10 mm — the most common size, compatible with standard 0° laparoscopes
- 12 mm — for larger instrument access with simultaneous visualization
Standard cannula lengths (typically 100 mm) are commonly used for abdominal laparoscopic access. Configurations may vary by manufacturer, market, and OEM specification requirements.
Single-Use Optical Trocars for Infection Control
Many optical trocars in current global procurement programs are supplied as single-use, disposable devices. Single-use optical trocars are typically:
- EO sterilized and individually sealed for ready-to-use deployment
- Supplied with full sterility documentation and traceability labeling
- CE MDR and ISO 13485 compliant where required by market regulations
- Available with private label and OEM packaging programs
For a broader overview of single-use vs reusable considerations across surgical consumables categories, see: Disposable vs Reusable Surgical Consumables — Procurement Comparison for Global Buyers .
Optical Trocar Procurement and OEM Sourcing Considerations
For medical device distributors and private label brands sourcing optical trocars through OEM or ODM programs, key evaluation criteria typically include:
Procurement Checklist — Optical Trocars
- Obturator transparency quality — optical clarity must support reliable scope visualization
- Scope compatibility — confirm laparoscope diameter fit (typically 5mm or 10mm scope channel)
- Cannula diameter and length options — 5mm / 10mm / 12mm availability
- Valve system and gas seal integrity — critical for maintaining pneumoperitoneum
- Sterilization method and documentation — EO sterilization, ISO 11135 compliance
- Regulatory certifications — CE MDR, ISO 13485, CFDA/NMPA as required by target market
- OEM / private label capability — packaging customization, SKU management
- Production consistency and supply stability — especially for high-volume distributor programs
Optical trocars are frequently sourced as part of a broader Trocar Essentials product program that includes classic, bladeless, and optical configurations — allowing distributors to consolidate trocar SKUs with a single manufacturing partner.
Why Source Optical Trocars from Senmed Medical?
Senmed Medical is a certified manufacturer of disposable laparoscopic surgical consumables with over 20 years of OEM and ODM manufacturing experience. Our trocar program covers Classic, Bladeless, and Optical configurations — enabling distributors and private label brands to consolidate their entire trocar SKU portfolio with a single, verified supply partner.
✔ Certified Manufacturing
ISO 13485 quality management system, CE (MDR) and CFDA certifications, EO sterilization validated to ISO 11135. Full traceability from raw material to finished product.
✔ Cleanroom Production Facility
700m² Class 100,000 cleanroom for injection molding, assembly, and packaging — with in-house physical and microbiological laboratory quality control.
✔ Full OEM / ODM Capability
In-house R&D, DFM engineering, precision mold development, and customized packaging — supporting private label programs for distributors in the UK, US, Europe, Australia, and Asia.
✔ Complete Trocar Portfolio
Classic, Bladeless, and Optical trocar configurations available under a single supplier program — simplifying procurement, documentation, and supply chain management for global distributors.
View Optical Trocar Specifications
Download product details, certifications, and available configurations.
Frequently Asked Questions About Optical Trocars
What is the main difference between an optical trocar and a classic trocar?
The key difference is visualization. An optical trocar uses a transparent obturator that accommodates a laparoscope, allowing surgeons to see tissue layers in real time during insertion. A classic trocar uses a traditional blade tip with no visual confirmation during access.
What is the difference between an optical trocar and a bladeless trocar?
A bladeless trocar separates tissue without a blade but does not provide visual feedback during insertion. An optical trocar — which may use either a cutting or dilating obturator tip — adds real-time visualization by allowing a laparoscope to pass through the clear obturator during access.
What size laparoscope is used with an optical trocar?
A 0° laparoscope is typically used with optical trocars during insertion. The scope diameter must match the trocar's inner channel specification — commonly a 5mm or 10mm scope channel, depending on the trocar configuration selected.
Are optical trocars single-use or reusable?
Most optical trocars in current global procurement programs are supplied as single-use, disposable devices — EO sterilized and individually packaged — to align with infection control standards and reduce reprocessing requirements.
Can optical trocars be supplied through OEM or private label programs?
Yes. Optical trocars are commonly sourced through OEM and ODM manufacturing programs for global medical device distributors and private label brands, typically as part of a complete trocar portfolio that includes classic and bladeless configurations.
What certifications should I look for when sourcing optical trocars?
Key certifications for global procurement include CE MDR (European market), ISO 13485 (quality management system), ISO 11135 (EO sterilization validation), and CFDA/NMPA for China market access. Requirements vary by target distribution region.
Request an Optical Trocar Sample or OEM Consultation
Senmed Medical supports global medical device distributors and private label brands with OEM and ODM optical trocar programs — including product sampling, regulatory documentation, custom packaging, and scalable supply chain solutions.
- CE MDR & ISO 13485 certified production
- Classic, Bladeless, and Optical trocar — full portfolio from one supplier
- OEM / private label programs for UK, US, Europe, Australia, Asia markets
- EO sterilization with ISO 11135 validation
- Sample requests and DFM consultation available